Abstract
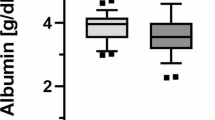
Changes in the antioxidant capacity of albumin and alterations of the albumin structural conformation were examined in patients in advanced stages of diabetes nephropathy. Human serum albumin was purified from diabetic patients in pre-dialysis (glomerular filtration rate [GFR] between 15 and 29 ml min−1 1.73 m−2) and those in dialysis (GFR ≤ 15 ml min−1 1.73 m−2) and then compared with albumin from patients with a normal GFR (>90 ml min−1 m−2). We evaluated the antioxidant capacity of albumin using an enhanced chemiluminescence-based assay and thiol group content, and the structural changes were evaluated by circular dichroism and fluorescence spectroscopy. The antioxidant capacity and thiol content of albumin from patients in advanced stages of diabetic nephropathy were markedly reduced. The circular dichroism spectra showed a mean albumin α-helix content reduction from 44 to 37 % and from 44 to 30 % between the control group and pre-dialysis and dialysis patients, respectively. Additionally, the fluorescence intensity was reduced by 4.2 and 13 % for the groups 4 and 5, respectively, in relation with the control. These data provide evidence for the partial denaturation of albumin and exacerbated oxidative stress among patients in advanced stages of diabetes nephropathy before and even after dialysis.



Similar content being viewed by others
References
Zelmanovitz T, Gerchman F, Balthazar AS, Thomazelli FCS, Jorge D, Matos JD, Canani LH (2009) Diabetic nephropathy. Diabetol Metab Syndr 1:1–17
Calabrese V, Mancuso C, Sapienza M, Puleo E, Calafato S, Cornelius C, Finocchiaro M, Mangiameli A, Di Mauro M, Stella AMG, Castellino P (2007) Oxidative stress and cellular stress response in diabetic nephropathy. Cell Stress Chaperones 12:299–306
Halliwell B, Whiteman M (2004) Measuring reactive species and oxidative damage in vivo and in cell culture: how should you do it and what do the results mean. Brit J Pharmacol 142:231–255
Roche M, Rondeau P, Singh NR, Tarnus E, Bourdon E (2008) The antioxidant properties of serum albumin. FEBS Lett 582:1783–1787
Otagiri M, Chuang VT (2009) Pharmaceutically important pre- and posttranslational modifications on human serum albumin. Biol Pharm Bull 32:527–534
Christodoulou J, Sadler PJ, Tucker A (1994) A new structural transition of serum albumin dependent on the state of Cys34. Eur J Biochem 225:363–368
Varshney A, Rehan M, Subbarao N, Rabbani G, Khan RH (2011) Elimination of endogenous toxin, creatinine from blood plasma depends on albumin conformation: site specific uremic toxicity and impaired drug binding. PLoS ONE 6:1–15
Oettl K, Stauber RE (2007) Physiological and pathological changes in the redox state of human serum albumin critically influence its binding properties. Br J Pharmacol 151:580–590
Turell L, Carballal S, Botti H, Radi R, Alvarez B (2009) Oxidation of the albumin thiol to sulfenic acid and its implications in the intravascular compartment. Braz J Med Biol Res 42:305–311
Himmelfarb J, McMonagle E (2001) Albumin is the major plasma protein target of oxidant stress in uremia. Kidney Int 60:358–363
Faure P, Tamisier R, Baguet JP, Favier A, Halimi S, Levy PE, Pepin J (2008) Impairment of serum albumin antioxidant properties in obstructive sleep apnoea syndrome. Eur Respir J 31:1046–1053
Watanabe A, Matsuzaki S, Moriwaki H, Suzuki K, Nishiguchi S (2004) Problems in serum albumin measurement and clinical significance of albumin microheterogeneity in cirrhotics. Nutrition 20:351–357
Faure P, Troncy L, Lecomte Wiernsperger MN, Lagarde M, Ruggiero D, Halimi S (2005) Albumin antioxidant capacity is modified by methylglyoxal. Diabetes Metab 31:169–177
Terawaki H, Era S, Nakayama M, Hosoya T (2011) Decrease in reduced-form albumin among chronic kidney disease patients: new insights in cardiovascular complications. Ther Apher Dial 15:156–160
Matsuyama Y, Terawaki H, Terada T, Era S (2009) Albumin thiol oxidation and serum protein carbonyl formation are progressively enhanced with advancing stages of chronic kidney disease. Clin Exp Nephrol 13:308–315
Taverna M, Marie AL, Mira JP, Guidet B (2013) Specific antioxidant properties of human serum albumin. Ann Intensive Care 3:4
Cohen MP (2003) Intervention strategies to prevent pathogenetic effects of glycated albumin. Arch Biochem Biophys 419:25–30
Rondeau P, Armenta S, Caillens H, Chesne S, Bourdon E (2007) Assessment of temperature effects on beta-aggregation of native and glycated albumin by FTIR spectroscopy and PAGE: relations between structural changes and antioxidant properties. Arch Biochem Biophys 460:141–150
Wratten ML, Sereni L, Tetta C (2001) Oxidation of albumin is enhanced in the presence of uremic toxins. Ren Fail 23:563–571
Soejima A, Kaneda F, Manno S, Matsuzawa N, Kouji H, Nagasawa T, Era S, Takakuwa Y (2002) Useful markers for detecting decreased serum antioxidant activity in hemodialysis patients. Am J Kidney Dis 39:1040–1046
American Diabetes Association (2013) Standards of medical care in diabetes–2013. Diabetes Care 36:S11–S66
Riener CK, Kada G, Gruber HJ (2002) Quick measurement of protein sulfhydryls with Ellman’s reagent and with 4, 4′-dithiodipyridine. Anal Bioanal Chem 373:266–276
Medina-Navarro R, Durán-Reyes G, Díaz-Flores M, Vilar-Rojas C (2010) Protein antioxidant response to the stress and relationship between molecular structure and antioxidant function. PLoS ONE 5:1–10
Moreno SN, Stolze K, Janzen EG, Mason RP (1998) Oxidation of cyanide to the cyanyl radical by peroxidase/H2O2 systems as determined by spin trapping. Arch Biochem Biophys 265:267–271
Halliwell B, Gutteridge JM (1999) Antioxidant defenses. Free radicals in biology and medicine, 3rd edn. Oxford University Press, New York, pp 105–245
Kalyanaraman B, Janzen EG, Mason RP (1985) Spin trapping of the azidyl radical in azide/catalase/H2O2 and various azide/peroxidase/H2O2 peroxidizing systems. J Biol Chem 260:4003–4006
Chapple I, Mason GI, Garner I, Matthews JB, Thorpe GH, Maxwell SR, Whitehead TP (1997) Enhanced chemiluminescent assay for measuring the total antioxidant capacity of serum, saliva and crevicular fluid. Ann Clin Biochem 34:412–421
Cockcroft DW, Gault MH (1976) Prediction of creatinine clearance from serum creatinine. Nephron 16:31–41
Pace CN, Vajdos F, Fee L, Grimsley G, Gray T (1995) How to measure and predict the molar absorption coefficient of a protein. Protein Sci 4:2411–2423
Peterman BF, Laidler KJ (1980) Study of reactivity of tryptophan residues in serum albumins and lysozyme by N-bromosuccinamide fluorescence quenching. Arch Biochem Biophys 199:158–164
Medina-Navarro R, Corona-Candelas I, Barajas-González S, Díaz-Flores M, Durán-Reyes G (2014) Albumin antioxidant response to stress in diabetic nephropathy progression. PLoS ONE 9:1–13
Kawakami A, Kubota K, Yamada N, Tagami U, Takehana K, Sonaka I, Suzuki E, Hirayama K (2006) Identification and characterization of oxidized human serum albumin. FEBS J 273:3346–3357
Dockal M, Carter DC, Ruker F (1999) The three recombinant domains of human serum albumin. J Biol Chem 274:29303–29310
Curry S (2009) Lessons from the crystallographic analysis of small molecule binding to human serum albumin. Drug Metab Pharm 24:342–357
Sugio S, Kashima A, Mochizuki S, Noda M, Kobayashi K (1999) Crystal structure of human serum albumin at 2.5 Å resolution. Protein Eng 12:439–446
Greenfield NJ (2006) Analysis of the kinetics of folding of proteins and peptides using circular dichroism. Nat Protoc 1:2891–2899
Sattarahmady N, Moosavi-Movahedi AA, Habibi-Rezaei M, Ahmadian S, Saboury AA, Heli H, Sheibani N (2008) Detergency effect of nanofibrillar amyloid formation on glycation of human serum albumin. Carbohydr Res 343:2229–2234
Kisugi RT, Kouzuma T, Yamamoto S, Akizuki H, Miyamoto Y, Someya J, Yokoyama I, Abe N, Hirai A, Ohnishi A (2007) Structural and glycation site changes of albumin in diabetic patient with very high glycated albumin. Clin Chim Acta 382:59–64
Nagaoka S, Cowger ML (1978) Interaction of bilirubin with lipids studied by fluorescence quenching method. J Biol Chem 253:2005–2011
Guerin-Dubourg A, Catan AA, Bourdon E, Rondeau P (2012) Structural modifications of human albumin in diabetes. Diabetes Metab 38:171–178
Zoellner H, Hou JY, Hochgrebe T, Poljak A, Duncan MW, Golding J, Henderson T, Lynch G (2001) Fluorometric and mass spectrometric analysis of nonenzymatic glycosylated albumin. Biochem Biophys Res Commun 248:83–89
Zoellner H, Siddiqui S, Kelly E, Medbury H (2009) The anti-apoptotic activity of albumin for endothelium is inhibited by advanced glycation end products restricting intramolecular movement. Cell Mol Biol Lett 14:575–586
Acknowledgments
This study was supported by the Fondo de Investigación en Salud FIS/IMSS/PROT/G10/854 and FIS/IMSS/PROT/896/Consejo Nacional de Ciencia y Tecnología (CONACYT) SALUD-2010-01-141937.
Conflict of Interest
The authors have no conflicts of interest associated with the content of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rosas-Díaz, M., Camarillo-Cadena, M., Hernández-Arana, A. et al. Antioxidant capacity and structural changes of human serum albumin from patients in advanced stages of diabetic nephropathy and the effect of the dialysis. Mol Cell Biochem 404, 193–201 (2015). https://doi.org/10.1007/s11010-015-2378-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2378-2